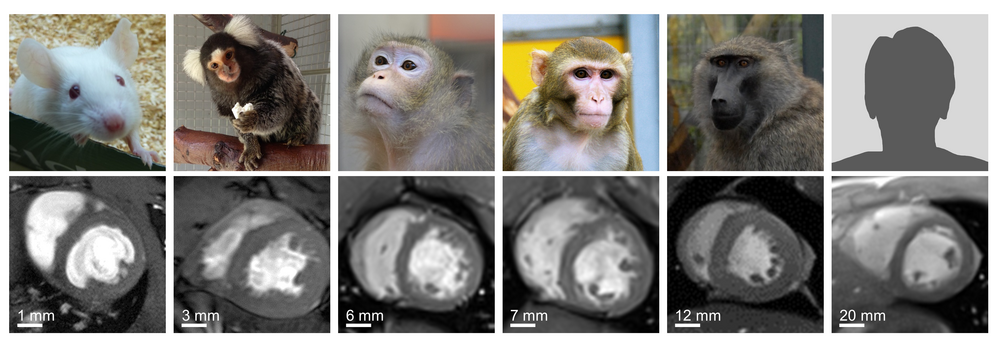
Quantitative Cardiovascular MR in non-human Primates
Cardiovascular diseases (CVD) are the leading causes of death worldwide. Translating novel treatment approaches into clinical application requires animal models resembling humans. In particular, non-human primates (NHPs), as our closest genetic relatives, are a most valuable model system for these translational studies.
However, monitoring treatment effects and disease progression requires a robust and reliable assessment of cardiac morphology and function. Magnetic Resonance Imaging (MRI), a non-invasive imaging modality, offers a unique opportunity to longitudinally monitor the remodeling process of the heart during translational studies. The main research focus of our group is to establish and develop quantitative methods for functional and metabolic cardiovascular MR in NHPs. The newly implemented methods have already been applied in a variety of research projects:
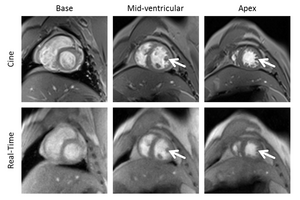
Reducing potential risk and burden of cardiovascular MR in NHPs using real-time cardiac MRI
The gold standard for non-invasive characterization of CVD in clinical routine is cine MRI, an ECG-gated imaging technique routinely acquired during periods of breath-holding. In NHPs, however, breath-holding has to be forced by artificial ventilation under anesthesia which significantly prolongs the measurement time with an additional risk for the animal. A promising alternative technology that has recently been introduced for imaging in humans is the so called real-time technique. Here, as achieved by an enormous acceleration of data acquisition combined with special image reconstruction methods, the formerly required breath-holding and ECG-gating can be discarded. This approach has now, for the first time, successfully been transferred to the smaller and much faster beating heart of macaques.
Longitudinal monitoring of myocardial remodeling
Progression of CVD is often associated with myocardial remodeling. This includes the accumulation of collagen in the extracellular matrix (localized or diffuse fibrosis), alterations of the cardiac energy metabolism (decreased fatty acid oxidation), cardiomyocyte hypo- or hypertrophy, and decreased vascular volume. In this research project we aim to individually analyze and distangle these distinct components of the myocardial remodeling process by a multi-compartment approach including structural, functional and metabolic imaging. Exemplarily, the assessment of structural (myocardial wall thickness and extracellular volume), functional (ventricular volumes and ejection fraction) and metabolic parameter (ATP-kinase) have been established in various species (baboons, macaques, marmosets, rabbits and mice) and have been successfully applied in different translational studies (stem cell therapy, aging, valve replacement, extracorporeal circulation). For example, in collaborative studies with the University Medical Center Göttingen and the Hannover Medical School we evaluate the safety and efficiency of stem cells-based approaches for the treatment of myocardial infarction.
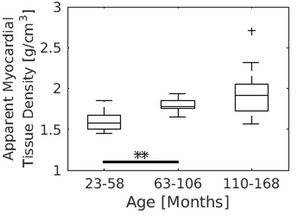
Marmosets as a primate model of cardiovascular aging
Given the continuously increasing life expectancy and the challenges arising from the associated increase in cardiovascular disease for the public health system, NHPs, including marmosets, have gained particular interest as potential model systems in the research of aging and age-related disease. In a joint study together with researchers from the Unit of Infections Models and the Platform Degenerative Diseases we could, for the first time, present quantitative MR parameters of cardiac function across different ages from young adult, middle-aged, to very old clinically healthy marmosets. Interestingly, significant age-dependent changes could be observed in the myocardial mass and the ventricular volume while parameters of cardiac function, such as ejection fraction and wall thickening, appeared almost stable with advancing age. A subsequent study in collaboration with the Laboratory Animal Science Unit currently focuses on potential, more subtle changes of cardiac function using mild challenges of the heart by pharmacological intervention and MR methods particularly suitable for imaging of cardiac fibrosis.
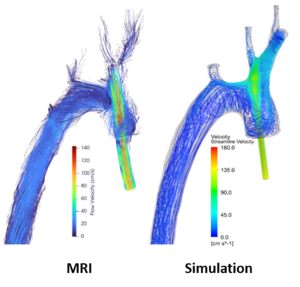
Analysis of the aortic blood flow in an animal model of extracorporeal circulation
Extracorporeal circulation (ECC) is routinely used in cardiac surgery and in patients with heart or lung failure to temporarily replace or support the restricted cardiopulmonary function. Possible damaging effects of ECC on the aortic blood flow and wall shear stress are well known and a variety of numerical simulations have been introduced to mimic the aortic blood flow. Nevertheless, a preclinical animal model that resembles realistic ECC conditions is essential to validate the outcome of these simulations. In a collaborative study with the Medical Faculty of the University of Düsseldorf and Department of Cardiovascular Engineering of RWTH Aachen, we are, for the first time, investigating the impact of different ECC scenarios on the aortic blood flow and cerebral perfusion in a realistic and reproducible animal model.