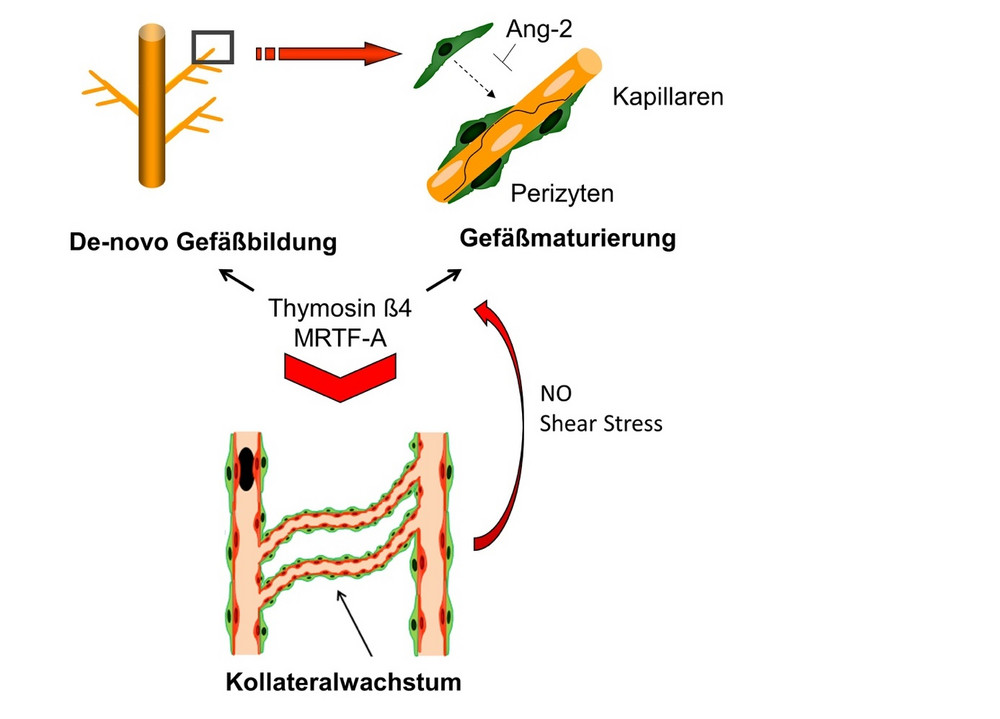
The gradual occlusion of coronary arteries can result in reversible loss of cardiomyocyte function (hibernating myocardium). This hibernating myocardium appears to be amenable to therapeutic neovascularization because it can be reactivated by adequate perfusion. However for therapeutic neovascularization in addition to angiogenesis, the maturation of these newly formed vessels is indispensable in our eyes. The two novel factors, thymosin ß4 and MRTF-A, are able, besides the induction of neovascularization, so called angiogenesis, to recruit significantly more pericytes. The essential role of vascular maturation was demonstrated in the rabbit hindlimb ischemia model. Whether this concept could be a therapeutic option in patients with ischemic cardiomyopathy was further investigated in a pre-clinical model of hibernating myocardium.
In this model, both thymosin ß4 and MRTF-A were able to enhance angiogenesis and arteriogenesis as well as perfusion in the myocardium. This increased perfusion significantly improved global and regional myocardial dysfunction. In summary, thymosin ß4 in cooperation with MRTF-A enhances angiogenesis by CCN-1 as well as vessel maturation by CCN-2, thus enabling an improvement of perfusion and function in the ischemic muscles.