The standard therapy in closed coronary vessels is a rapid recanalization of the affected vessels, the so-called revascularization. This method is time-sensitive and the proportion of rescued tissue decreases the longer the vessel occlusion lasts. However, the reopening of the closed vessel also causes damage, the so-called reperfusion damage. This is caused by the rapid influx of oxygen-rich blood into the ischemic heart tissue and is characterized by increased cell death, immigration of inflammatory cells and reduced contractile performance in this area. This damage is partly reversible and therefore very suitable for new therapeutic strategies. Optimized pharmacological and interventional therapy has been shown to significantly improve the survival of patients with myocardial ischemia, but in particular in patients with major myocardial infarction, adverse remodeling occurs, what can lead to heart failure in a high number of patients (30% in the AMI group).
Akuter Myokardinfarkt und Revaskularisierung
Therapy approaches
MicroRNAs (miRs) are small, approximately 22 nucleotide long, noncoding RNAs that are processed intracellularly by the RNases Drosha and Dicer. By binding to mRNAs, miRs can prevent translation or induce mRNA degradation and thus regulate a variety of target genes posttranscriptionally. MiRs play a central role in developmental biology, in the maintenance of tissue homeostasis and in particular under pathophysiological conditions. Various studies have shown that the pharmacological intervention with miR antisense molecules can be used to specifically reduce miR in animal models.
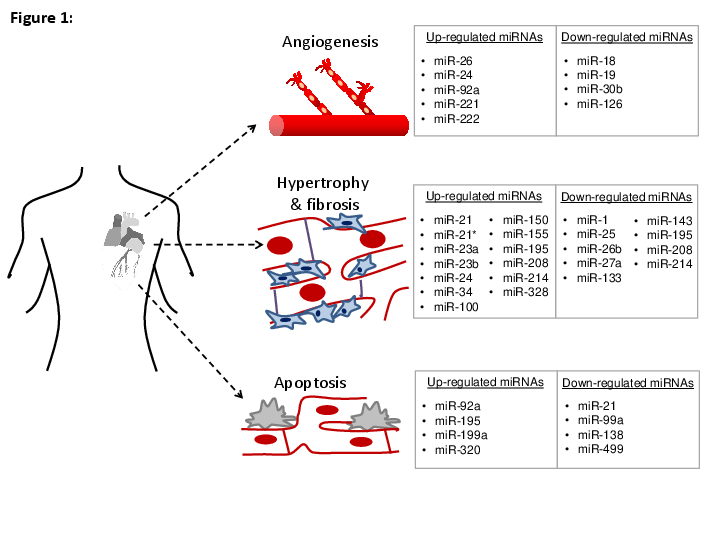
The inhibition of miR-92a by means of an LNA-modified antagonist showed a marked improvement in ischemia-reperfusion damage. In addition to reducing infarct size, this has shown an improvement in microcirculation and a reduction in local inflammation, all key processes involved in ischemia-reperfusion injury.
