Why are genetically modified animals needed for research purposes?
Mammals have developed over evolutionary periods from common ancestors. Thus, the basic physical structure and functions of their organs are very similar. For example, they all have a basically identically structured heart or a central nervous system that processes sensory perceptions and coordinates movements. Many general aspects of the mechanics of mammals, including that of humans, could therefore be studied in any mammalian species and in the process learn a lot more of the functions of the human body. Rats and mice are the most commonly used animal models in biomedical research and are therefore also responsible for most of the groundbreaking findings in medical research. A specific benefit of research with mice is that it has been possible for more than 30 years to selectively change their genomes; to make them transgenic or in general to modify them genetically.
![Diagram of a gene in the genomic DNA. Image: Thomas Splettstoesser (www.scistyle.com), [CC BY 4.0 (http://creativecommons.org/licenses/by/4.0)], Wikimedia Commons](/fileadmin/_processed_/2/a/csm_Chromosom-DNA-Gen_032400cff2.png)
Hereditary diseases are genetic
A substantial part of the genetic material (the genome) is organized in genes. Genes are specific sections in the genome that contain the information for a highly essential group of molecular building blocks (proteins) of the body. The human genome has approximately 23,000 genes. It is known, that certain diseases are hereditary and such diseases are referred to as genetic diseases. They stem from a gene malfunction but more specifically that of its protein whose production information is stored in the corresponding genes.
Incurable genetic diseases include diseases such as Huntington’s disease or leukodystrophy. More frequent and also serious diseases such as Parkinson's disease or cardiac failure can also have genetic causes. Due to molecular biological and technical innovations, the diagnosis of the genetic causes of many hereditary has been possible for a number of years. In the meantime, it is commonly known that the loss of the function of certain genes, or the associated proteins, or even a specifically modified function of a protein, can trigger the diseases.
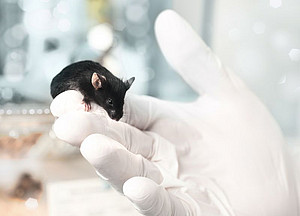
Unfortunately, a cure for many serious diseases is still to be found. This is mainly due to the fact that the causes of a disease in humans can be described, but we do not understand their effects on the cells, organs and the organism. The main problem is often the unavailability of the relevant examination material, e.g. the afflicted tissue is not obtainable. It is however, for ethical and medical reasons not permitted to analyze brain tissue sample of Parkinson's patients. This prevents a better functional understanding of the origin of many diseases and also the development and testing of new therapies. This is why a large gap currently exists between the genetic and molecular biological diagnosis of diseases and the understanding of the functional effects of these genetic changes in an entire organism, e.g. in human beings as patients. This functional understanding is of crucial importance for the development of a therapy.
This gap can now be partially filled by the development and broader application of stem cell and organoid models. Organoids in particular can provide good insights into some aspects of the function of real organs, but even they are still far from an organ embedded in a whole organism in terms of their biological complexity and regulation. However, stem cell and organoid models have the advantage that they can be made with human cells. Although not at the organismic level, human stem cell and organoid models better reflect the human situation at the molecular and cellular level than animal models. Thus, together with animal models, they are very useful for researching diseases and developing therapies.
Mice have very many genes that also occur in the human genome. Nevertheless, the corresponding genetic diseases that humans often suffer from are not always observed in animals. (This is probably not because these genetic diseases never occur in mice, but because these animals suffering from such diseases - unlike humans under medical treatment - simply do not survive in nature). However, the targeted switching off or changing of genes in the mouse can often reflect a disease of a human. Thus, it is a very helpful approach to simulate in mice the genetic causes of diseases diagnosed in humans, e.g. to generate transgenic or genetically modified mice and to obtain tissue samples for a better understanding of the cell biological and organismic causes of diseases. It is also a helpful approach to right from the start test new therapies, where the desired positive effects as well as possible side effects can be assessed.

Why is it important to have genetically modified primates?
There are genes in the human genome that are not found in mice. These additional genes, which have developed only in the course of primate development, are not found in the genome of other mammals. They are primate-specific, and are only found in monkeys, apes and humans. In the course of evolution, some human genes with a fundamental correspondence to mice, have acquired additional properties. Newly developed and slightly modified genes allow us, for example, to distinguish ourselves from other mammalian groups as primates. Nobody can argue the fact that we clearly differ from mice. Be it something as simple as measuring the pulse rate of a human at about 60 to 80 beats per minute in comparison to that of mice at 600 to 700 beats per minute. Or the lifespan: 70 to 90 years compared to one to two years. And also certain cognitive abilities that are only found in human beings. These differences also illustrate the limitations of mice when it comes to certain diseases that are human-specific and therefore not found in other mammals.
Here, it can be of an advantage to in addition to mice, use animals that are evolutionary closer to humans as models. Monkeys have a genome comparable to humans, and their collective bodily function is similar to that of humans. It is probable that some diseases that cannot be depicted in mice through genetic modification can be depicted in monkeys. Such genetically modified monkeys could be a major step toward a better understanding of very significant, previously incurable and fatal diseases and thus bringing new therapies within reach.
The following must however be considered:
- Nobody can nowadays safely predict whether and to what extent genetically modified monkeys will actually contribute to the development of therapies. It is a rational and bio-medically justifiable approach that is likely to bring important new insights into severe human diseases.
- Gained knowledge and therapy development based on genetically modified animals in general and specifically monkeys can only be expected in the long-term. Realistic time-frames should be decades rather than years. In view of the potential importance of transgenic primates, Japan, China and the USA have begun extensive work in this field, with the aim of becoming an international biomedical leader.
- Genetically modified animals used as models for serious and fatal human diseases will at some point be euthanized. This time-point must be determined in order to minimize the suffering of the animals in the same way that the suffering of patients for whom the research is conducted, is minimized. The intention should be, not to let the animals reach a point where they lose their sense of orientation such as in Alzheimer patients or completely lose their ability to move such as in severe progression of Parkinson’s disease. Simulation of earlier phases of the disease is from a research point of view, far more important because it enables the investigation of the crucial molecular and cell biological as well as the early functional changes that led to the development of a disease. In addition, it is much more useful to test therapy approaches at an early stage, as therapies often prevent progress and a reversal is not possible. It is not expected of the animals to suffer in the same way that the patients do.
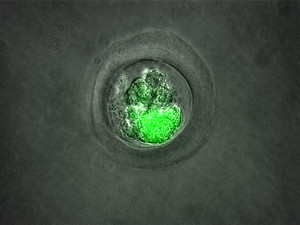
Transgenic non-human primates at the DPZ
At DPZ, the methods for genetic modification of comon marmosets by insertion of the gene for the pathologically inactive protein Green Fluorescent Protein have been established. Currently, work is being done on the genetic modification of common marmosets using the CRISP/Cas system. This method allows the targeted modification of the monkey genome and has been tested for its accuracy in cell cultures. The aim of the current work at the DPZ is to use a genetically modified deaf monkey to preclinically test a gene therapy for genetically induced deafness in humans for its safety and efficacy.
Contact

Prof. Dr. Rüdiger Behr Head of Platform Degenerative Diseases +49 551 3851 132 +49 551 3851 431 Contact
Transgenic Common marmosets
The video shows a common marmoset family at the DPZ’s Primate Husbandry. Some of the animals carry the GFP transgene, which they have received from the mother, and some have an unchanged genetic makeup. There is no visible difference between the transgenic and non-transgenic animals.
