The fight against HIV / AIDS
A vaccine against the retrovirus HIV that was first discovered in the 1980s is yet to be found and the virus-induced syndrome of immune deficiency AIDS is still not curable. According to the WHO, around 36 million people are infected with HIV. The need for research in this area is therefore unquestionably urgent. It has now been scientifically established that the various variants of HIV that are currently known, were transferred from monkeys to humans.
The sources for the human immunodeficiency virus are probably different: Mangabeys, chimpanzees and gorillas in different regions of Africa is where researchers found original hosts of various viruses from which the human HIV strains have developed. Since many non-human primate species can get similar retroviral diseases, these animals are the best suited to investigate the virus. In certain Asian macaque species such as rhesus macaques (Macaca mulatta), the "Simian Immunodeficiency Virus" (SIV) triggers a disease that is very similar to AIDS. In addition, scientists use a virus hybrid from monkey and human viruses called SHIV in the primate model. Read more about HIV in our Info Center.
Animal experiments with non-human primates have always played an important role in HIV research. Although a large number of volunteers could be found for the testing of new drugs because of the severity of the disease, a lot of fundamental research was needed to elucidate the pathogenesis, the course of infection, and the adaptability of the HIV virus for which scientists had to rely on primate models. In the US Center for Disease Control (CDC) early findings on the disease progression of AIDS were obtained with the aid of infected common marmosets (Callithrix jacchus). In the early 1990s Animal experiments with chimpanzees were necessary in order to develop a blood test, with which HIV infection could be clearly verified.
In HIV research, macaques are very often used to test antiviral drugs. Since the controlled infection with HIV for the examination of the infection course is for ethical and legal reasons not allowed for humans, the animals are vital for the testing of prophylaxis agents. A special case of prophylaxis research, in which monkeys have played an indispensable role as models, is research on the mother-to-fetus transmission of the virus. Test series for the best protection of the unborn from the infection was of course only possible with non-human primates. The findings from such studies are then incorporated into the anti-retroviral treatment of pregnant women.
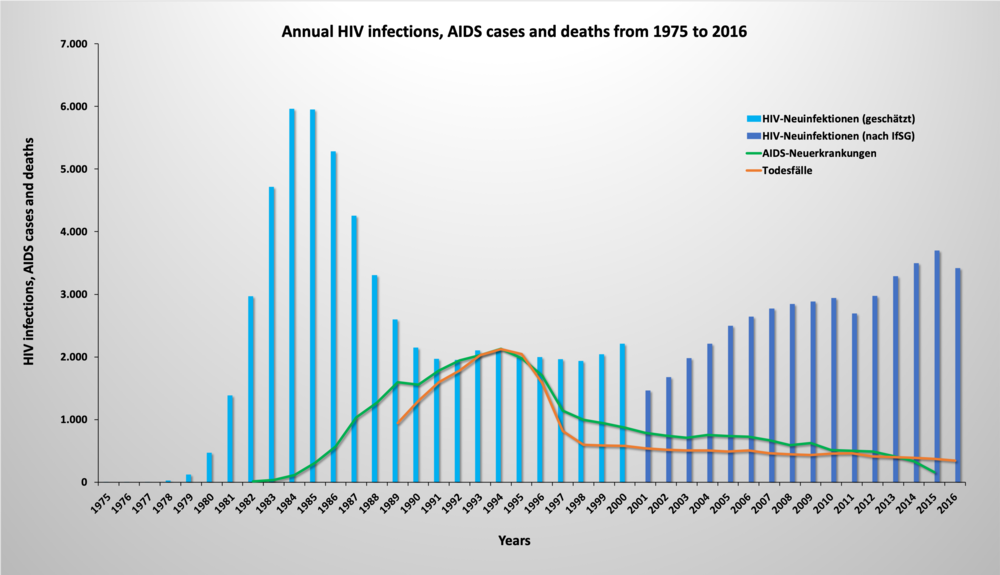
The chart above shows the number of new HIV infections, new AIDS cases and deaths from 1975 to 2016. The data on new HIV infections up to the year 2000 are based on estimates by the Robert Koch Institute, and from 2001 on the figures reported according to the German Infection Protection Act (IfSG). The number of HIV cases was highest in Germany in the mid-1980s. It decreased again in the second half of the 1980s and remained at a comparatively low level in the 1990s. An increase has been observed since 2001. While the number of AIDS cases and deaths initially rose from the end of the 1980s onwards, they gradually declined with the introduction of antiretroviral therapy (ART) in 1996. The treatment, which inhibits virus replication in the body through a combination of various drugs, enables HIV patients to significantly extend their life expectancy and thus lead an almost normal life. Graphic: Sylvia Ranneberg, Sources: Robert-Koch-Institut (Hrsg) (2014): Schätzung der Prävalenz und Inzidenz von HIV-Infektionen in Deutschland (Stand: Ende 2013). Epidemiologisches Bulletin 44:429-437; Database Health Monitoring of the Federal Government, infectious/notifiable diseases, HIV infections, AIDS cases, deaths, www.gbe-bund.de
Truvada – Prophylaxis and virus inhibitors
In July 2012, the FDA approved Truvada as a preventative measure for the American market. Truvada is used to reduce the risk of an infection. In various studies, scientists have shown that the drug can reduce the risk by at least 73 percent. In a study with homosexual men, an efficacy of up to 90 percent was given. The agent blocks the growth of the virus and has been used to delay the outbreak of AIDS. It should now be implemented to protect partners of infected from contracting the immunodeficiency virus.
Rhesus monkeys were used to test the effectiveness and to determine the dosage of the drug, based on tenofovir. Tenofovir is an antiviral that is used in the treatment of both HIV-1 and hepatitis B. Due to its structural similarity with a natural nucleotide, Tenofovir is incorporated into the genes of host cells that are infected with the virus and in doing so it subsequently inhibits the growth of the viruses in the body.
Macaques were used to test the toxicity of tenofovir. Tenofovir was approved in 2001 in the US and 2002 in the EU in the form of the tenofovir disoproxil fumarate as a tablet for the treatment of HIV infections in adults, but must always be used in combination with other antiretroviral medicinal products.
In the case of Truvada, test series with rhesus monkeys showed that the protective effect depends essentially on the regular intake: When the drug was administered three days before the infection, five out of six infected monkeys were protected and an intake of two weeks before infection showed that the protective effect was no longer detectable. With the help of drugs such as Truvada, infections can be prevented and infected persons can live with the virus for decades without great limitations. In the United States where there are approximately 50,000 newly infected patients each year, Truvada helps to reduce the rate of new infections. As reported by the Robert Koch Institute, a total of 3260 new diagnoses were made in Germany in 2013, an increase of ten percent compared to the previous year. According to the Robert Koch Institute, about 80,000 HIV patients live in Germany.
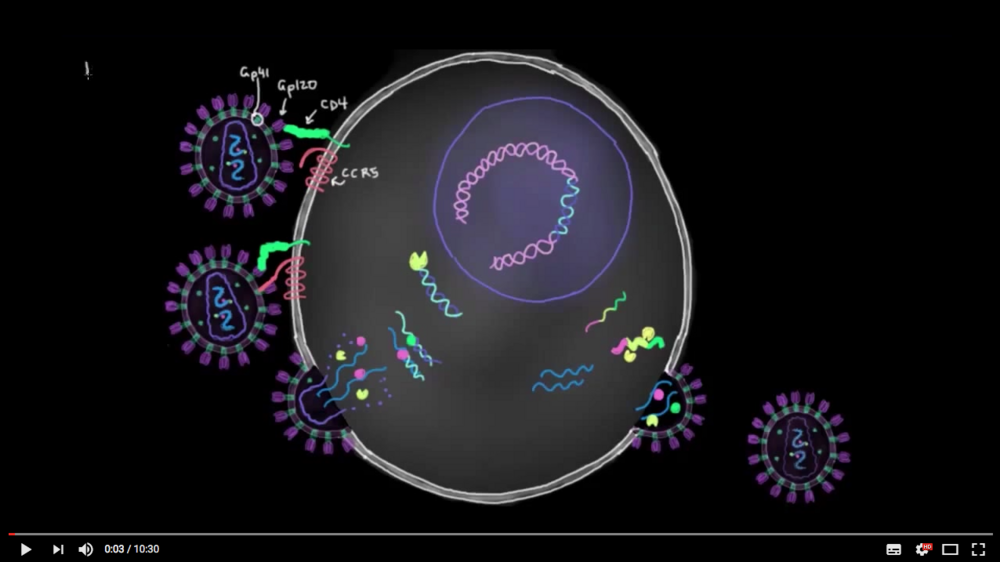
In this short video you will see how antiretroviral drugs that were developed through animal experiments on non-human primates work.