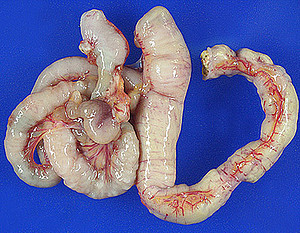
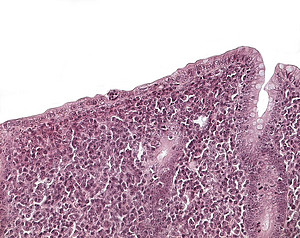
The Wasting Marmoset Syndrome (WMS) is a disease that affects captive callitrichids and causes substantial problems in zoological and experimental marmoset husbandry. So far pathogenetic mechanisms and etiologic factors of this complex syndrome could not be identified. The WMS is characterized by progressive weight loss despite of normal food intake and a deteriorating general condition. Chronic diarrhea, alopecia, muscle atrophy, chronic colitis as well as alterations of liver and kidneys are regarded as associated features of the disease. Recent studies about the WMS indicate that inflammatory intestinal changes play a decisive role in the pathogenesis of the disease. 23 common marmosets from two laboratory facilities (German Primate Center and a laboratory animal facility) with clinical signs of WMS were included in the present study. Postmortal light microscopic investigations with main focus on the gastrointestinal tract were carried out to evaluate grade and extent of the respective lesions. For phenotypic characterization of the intestinal inflammation the histologic examinations were completed by immunohistochemical techniques. Moreover clinical studies of live wasters, pathomorphologic investigations of the remaining organ systems and diverse etiologic analyses were carried out with the objective to define the WMS uniformely and to obtain indices about causal factors.
The results can be summarized as follows:
1. Weight development and therapeutic success were regarded as decisive diagnostic criteria for clinical evaluation of common marmosets with WMS. Adult common marmosets showed severe weight losses up to 45 % of the body weight, while subadult wasters were characterized by insufficient weight gain. Laboratory diagnostic and postmortal examinations were essential for the definite identification of a waster.
2. All common marmosets with WMS from the German Primate Center were adult animals that mainly fell ill at the age of 3 to 8 years. Body weights at necropsy varied between 230 g and 320 g. The common marmosets from the laboratory animal facility were predominantly classified as subadult and had body weights between 200 g and 260 g at the time of necropsy. A sex predisposition for the WMS was not existent.
3. Predominant organ changes were located in the intestinal tract, the kidneys and the liver. Chronic and chronic active enteritis was regarded as a decisive pathogenetic factor in terms of malassimilation. The other organ lesions presumably developed secondary in the course of disease.
4. Phenotypic characterization of the intestinal inflammation using immunohistochemical techniques resulted in a T-cell mediated local immune response.
5. Microbiological examinations were unsuggestive of infectious causes for the WMS. By means of pedigree analyses a monogenetic hereditary defect could be excluded. Thus it can be assumed that the WMS is a multifactorial disease with exogenous and endogenous contributing factors.
6. Comparative observation of the two marmoset facilities (German Primate Center and the the laboratory animal facility) revealed no decisive differences regarding the clinical nd pathomorphologic picture of the WMS.
Pathomorphologic investigation of WMS-diseased animals regularly revealed kidney alterations in terms of immune complex glomerulonephritis (ICGN) with mesangial proliferative character and chronic interstitial nephritis. Therfore, additional studies were performed on kidneys of 47 common marmosets suffering from WMS and 11 clinically healthy animals with special regard to histopathologic (paraffin and plastic embedding) and immunohistochemical methods (IgA,IgM, C3c-complement and α-sma). Distinct similarities between the ICGN in callitrichids and the human disease of IgA-nephropathy have been described previously. Therefore, ICGN was categorized in accordance to the classification of IgA-Nephropathy (IgAN) of humans proposed by HAAS (1997). The investigation revealed a high prevalence (98 %) for ICGN. Interestingly, apart from one marmoset, also all clinically healthy animals showed mesangial proliferative changes. The majority of common marmosets suffering from WMS showed a focal proliferative glomerulonephritis graduated as HAAS subclass III ICGN; subclass I was diagnosed in 4.2 %, subclass II in 19.1 %, subclass IV in 10.6 %, and subclass V 4.3 % of the cases. HAAS subclasses IV and V were mainly diagnosed in elderly animals with accompanying clinical manifestation. Transmission electron microscopy of selected cases revealed mesangial electron-dense deposits in proliferated glomeruli. There were no signs for origin- or sex-related predispositions for ICGN. In accordance to previous studies, immunohistochemical investigations revealed mesangial deposits of IgM in all animals. IgA could be detected in 47 % of all cases, whereas statistical results suggest a higher pathogenic potential of IgA compared to IgM. Common marmosets suffering from WMS showed significantly higher grades compared to the control group. Therefore, a certain positive correlation between WMS and ICGN seems likely. However, the occurrence of ICGN in clinically inconspicuous animals suggests a WMS-independent, multifactorial immunologic disease pattern.
Associated literature:
Renal lesion in common marmosets (Callithrix jacchus) with wasting marmoset syndrome