Immune cells with functional upgrade
Cytomegalovirus (CMV) belongs to the large family of herpes viruses and is distributed worldwide. According to the Robert Koch Institute, around 70 percent of adults in Germany are CMV-positive. Like other herpes viruses, CMV remains in the body for a lifetime after initial infection. In immune-healthy individuals, the disease is unproblematic and manifests itself, if at all, only with cold symptoms. However, life-threatening complications can occur in newborns and immunosuppressed individuals, resulting in pneumonia, liver and intestinal damage, and inflammation of the retina or even blindness. If pregnant women become infected with the virus, this can lead to growth retardation, hearing damage and neurological late damage of the child.
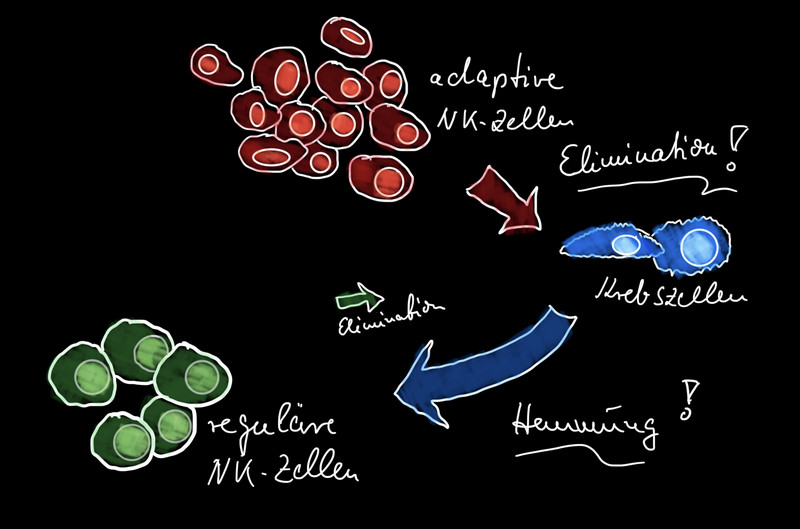
At the level of the immune system, it is known that CMV infection in humans can lead to increased production of special immune cells. These adaptive natural killer (NK) cells have a special receptor on their surface and can bind and kill CMV-infected cells. This is how our immune system controls the latent infection and keeps it at bay.
NK cells belong to the lymphocytes of the innate immune system. Their task is to eliminate virus-infected cells and tumor cells in the body. Adaptive natural killer cells have the advantage over regular NK cells that they react more quickly, are more durable and have a memory effect. About one-third of CMV-positive people have the adaptive NK cells in greater numbers.
A team of researchers led by Lutz Walter, head of the Primate Genetics Laboratory at the German Primate Center, wanted to find out whether the adaptive NK cells are also detectable in CMV-infected rhesus monkeys. Just as in humans, the virus is also widespread in this primate species.
"First, we produced antibodies with which we could specifically detect the receptor of the adaptive NK cells; this had not been possible before," says Lutz Walter. "Thus, we were able to identify the NK cells in question beyond doubt and were able to show that up to 73 percent of the NK cells in CMV-positive animals were adaptive cells. This means that the effect of CMV infection on the formation of adaptive NK cells works the same way in monkeys as it does in humans. So, the process apparently arose in primates a long time ago and is evolutionarily conserved."
The study thus shows that rhesus macaques are a valuable animal model for studying CMV infection in humans. In mice, for example, the immune response after CMV infection is different. Therefore, they are of limited use as experimental animals to study specific aspects of the disease.
An additional advantage of adaptive NK cells is that, unlike regular NK cells, they can also eliminate certain tumor cells via their special receptor. This antitumor effect makes them potentially interesting for new anticancer therapies. Further research into how adaptive NK cells function in primates is therefore important both for a better understanding of CMV infection and for cancer research.
Original publication
Hasan MZ, Höltermann C, Petersen B, Schrod A, Mätz-Rensing K, Kaul A, Salinas G, Dressel R, Walter L (2022): Detailed phenotypic and functional characterization of CMV-associated adaptive NK cells in rhesus macaques. Frontiers in Immunology 13: 1028788. doi: 10.3389/fimmu.2022.1028788, https://doi.org/10.3389/fimmu.2022.1028788