Do known drugs help against SARS-coronavirus-2?
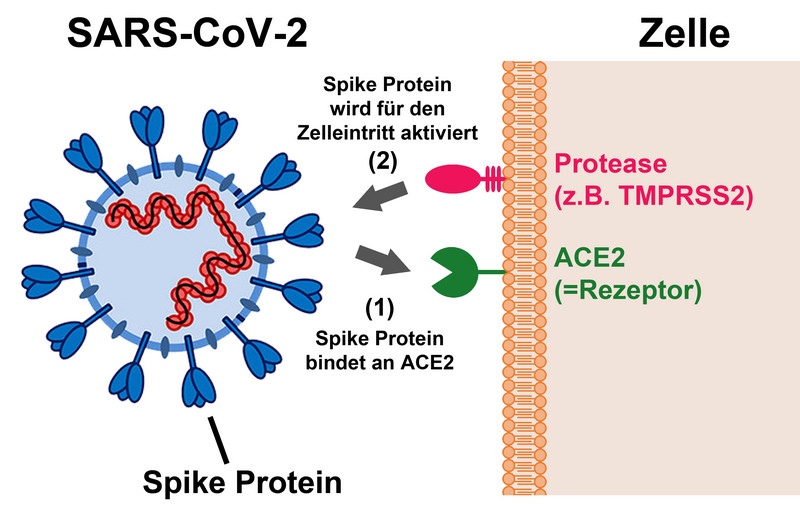
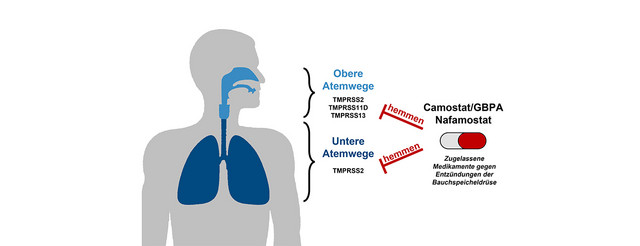
There are no therapeutics available that have been developed for COVID-19 treatment. Repurposing of already available medication for COVID-19 therapy is an attractive option to shorten the road to treatment development. The drug Camostat could be suitable. Camostat exerts antiviral activity by blocking the protease TMPRSS2, which is used by SARS-CoV-2 for entry into cells. However, it was previously unknown whether SARS-CoV-2 can use TMPRSS2-related proteases for cell entry and whether these proteases can be blocked by Camostat. Moreover, it was unclear whether metabolization of Camostat interferes with antiviral activity. An international team of researchers around Markus Hoffmann and Stefan Pöhlmann from the German Primate Center (DPZ) - Leibniz Institute for Primate Research has now shown that SARS-CoV-2 can use several TMPRSS2-related proteases for its activation. These proteases are expressed in the upper respiratory tract and are blocked by Camostat. In addition, the researchers found that Camostat and its major metabolite GBPA inhibit SARS-CoV-2 infection of primary human lung tissue. These findings support the further development of Camostat and related compounds for COVID-19 therapy (EBioMedicine).
SARS-CoV-2 depends on activation by the cellular protease TMPRSS2 for infection of lung cells. The researchers of the Infection Biology Unit of DPZ previously documented that the drugs camostat and nafamostat, which are used in Japan to treat inflammation of the pancreas, block SARS-CoV-2 infection by inhibiting TMPRSS2. However, it was unknown whether Camostat metabolites also block SARS-CoV-2 and whether the virus may use TMPRSS2-related proteases for infection that may be Camostat-insensitive.
An international team of researches around Markus Hoffmann and Stefan Pöhlmann from the Infection Biology Unit of the DPZ has now shown that SARS-CoV-2 can use several TMPRSS2-related proteases for infection, among them TMPRSS11D and TMPRSS13. These proteases may support viral spread in the upper respiratory tract and are blocked by Camostat. This finding indicates that switching to activators other than TMPRSS2 might not allow the virus to replicate in the presence of Camostat.
The researchers could show that not only Camostat but also a major Camostat-metabolite, GBPA, block TMPRSS2 and SARS-CoV-2 infection. "In the human body Camostat is rapidly converted to GBPA. Therefore, it was crucial to demonstrate that not only Camostat but also GBPA exert antiviral activity", says Stefan Pöhlmann, the head of the Infection Biology Unit of DPZ. Markus Hoffmann, the first author of the study adds: "Our results suggest that Camostat/GBPA may unfold antiviral activity in patients. However, for effective treatment of COVID-19, a higher Camostat dose might be required as compared to pancreatitis treatment."
Inhibition of SARS-CoV-2 by Camostat was initially shown using the lung cell line Calu-3. The participation of Prof. Armin Braun, Fraunhofer ITEM, Hannover, and Danny Jonigk, Institute of Pathology at the MHH, in the consortium allowed analysis of Camostat antiviral activity in primary human lung tissue ex vivo. Camostat and GBPA blocked SARS-CoV-2 infection of lung tissue and Nafamostat had an increased antiviral activity. Therefore, the team of the Infection Biology Unit and the laboratory of Armin Braun are investigating how Nafamostat can be directly delivered into the human lung for increased antiviral activity. This project receives financial support from the Bundesministerium für Bildung und Forschung (BMBF) (project RENACO, Repurposing of Nafamostatmesylat for treatment of COVID-19).
"Our results on the antiviral activity of Camostat and GBPA are relevant beyond the treatment of COVID-19. TMPRSS2 also plays an important role in other respiratory infections. Thus, Camostat could also be successfully used to treat influenza," says Markus Hoffmann.
Original publication
Hoffmann, M et al. (2021). Camostat mesylate inhibits SARS-CoV-2 activation by TMPRSS2-related proteases and its metabolite GBPA exerts antiviral activity. EBioMedicine, DOI: 10.1016/j.ebiom.2021.103255